|
Contributed by: New York State Education Department (NYSED)
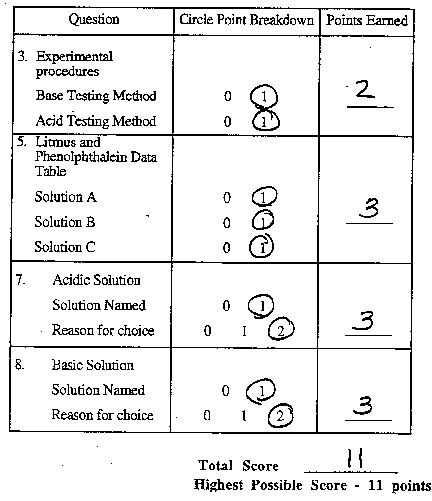
Here are examples of possible score points. (You will notice that
the questions in these examples are not phrased exactly as they
were in the task although the basic idea is the same. It is apparent
that the questions were edited post-field testing.)
|
Acid and Base Testing
Task: At this station, you will design and
carry out an experiment to determine which of three solutions
is acidic and which is basic.
|
MATERIALS:
- dropper bottle marked A
- dropper bottle marked B
- dropper bottled marked C
- dropper bottle with phenolphthalein
- blue litmus paper
- red litmus paper
- test card
- wax paper sheets
- waste containers
- paper towels
BACKGROUND:
- Phenolphthalein is a colorless indicator. When phenolphthalein
is added to a basic solution, the solution turns pink.
- Litmus paper is another indicator. Blue litmus paper turns red
(pink) when dipped in an acidic solution, which red litmus paper
turns blue when dipped in a basic solution.
DIRECTIONS:
- Using the information above, what will you do to determine which
solution(s) are acidic and which are basic? Using only the materials
listed above, outline the plan for your experiment in the space
below.

- CARRY OUT YOUR PLAN.
When carrying out your experiment, place a wax paper sheet over
the test card to protect it.
- Record your observations in the data table below.
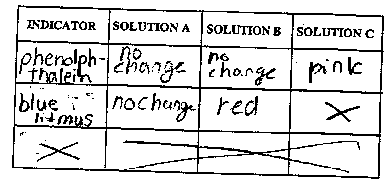
- Based on your observations, which solution is acidic?
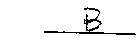
Explain the reason for your conclusion in the space below.

- Based on your observations, which solution is basic?
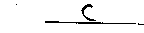
Explain the reason for your conclusion in the space below.

NYS Alternative Assessment in Science Project
Copyright, April 1996
The State University of New York
The State Education Department
Albany, New York 12234
|